 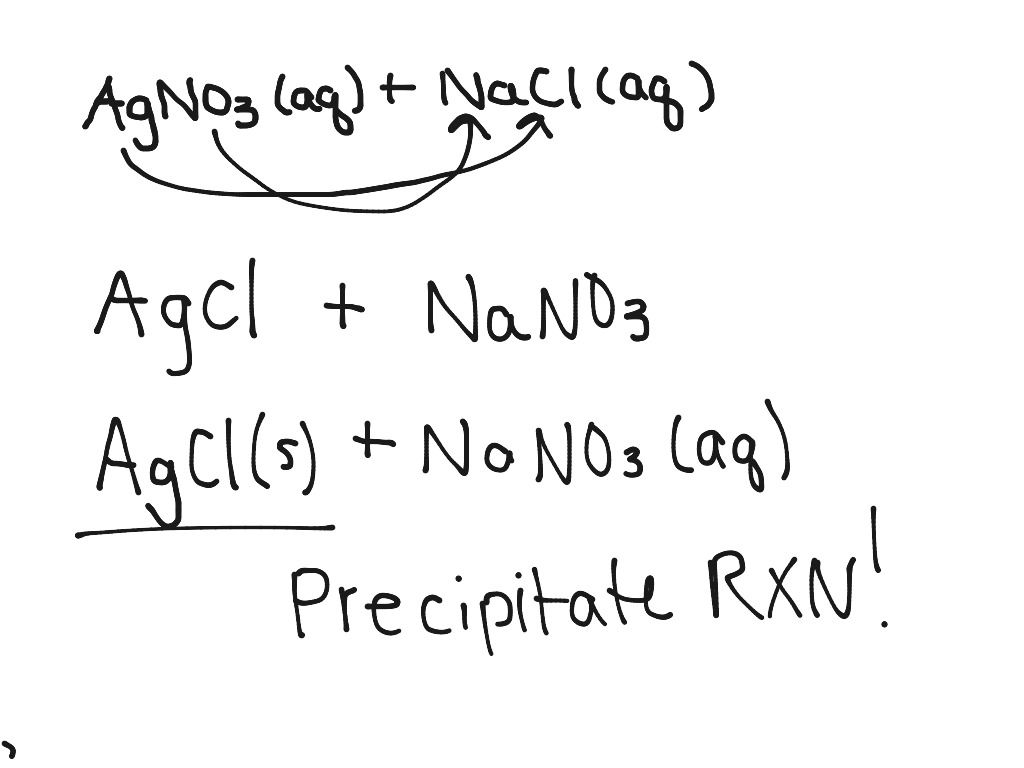
Explain the term reactant product giving examples. However, the same reaction occurs at a faster rate on adding manganese dioxide (MnO 2),Ĭ. How can the rate of the chemical reaction, namely, decomposition of hydrogen peroxide be increased?Īt room temperature, the decomposition of hydrogen peroxide into water and oxygen takes place slowly. Thus oxidation and reduction reactions occur simultaneously.ī. In this reaction, oxygen is removed from copper oxide therefore it is a reduction of CuO, while hydrogen accepts oxygen to form water that means oxidation of hydrogen takes place. In redox reaction, the reductant is oxidized by the oxidant and the oxidant is reduced by the In a redox reaction, one reactant gets oxidised while the other gets reduced during a reaction. The reaction which involves simultaneous oxidation and reduction is called an oxidation-reduction or redox reaction. What is the reaction called when oxidation and reduction take place simultaneously? Explain with one example. white precipitate is formed by exchange of ions Ba ++ and SO 4 - between the reactants.Ī. 
Addition of an aqueous solution of ZnSO 4 to an aqueous solution of BaCl 2 is an example of ……… reaction.Īddition of an aqueous solution of ZnSO 4 to an aqueous solution or BaCl 2 is an example or double displacement reaction.īarium chloride reacts with zinc sulphate to form a white precipitate of barium sulphate. This decomposition takes place with the help of an electric current, it is also called electrolytic decomposition.ĭ. When electric current is passed through acidulated water decomposition of water takes place. When electric current is passed through acidulated water ……. A process in which a metal or its ion loses one or more electrons is called an oxidation.Ĭ. 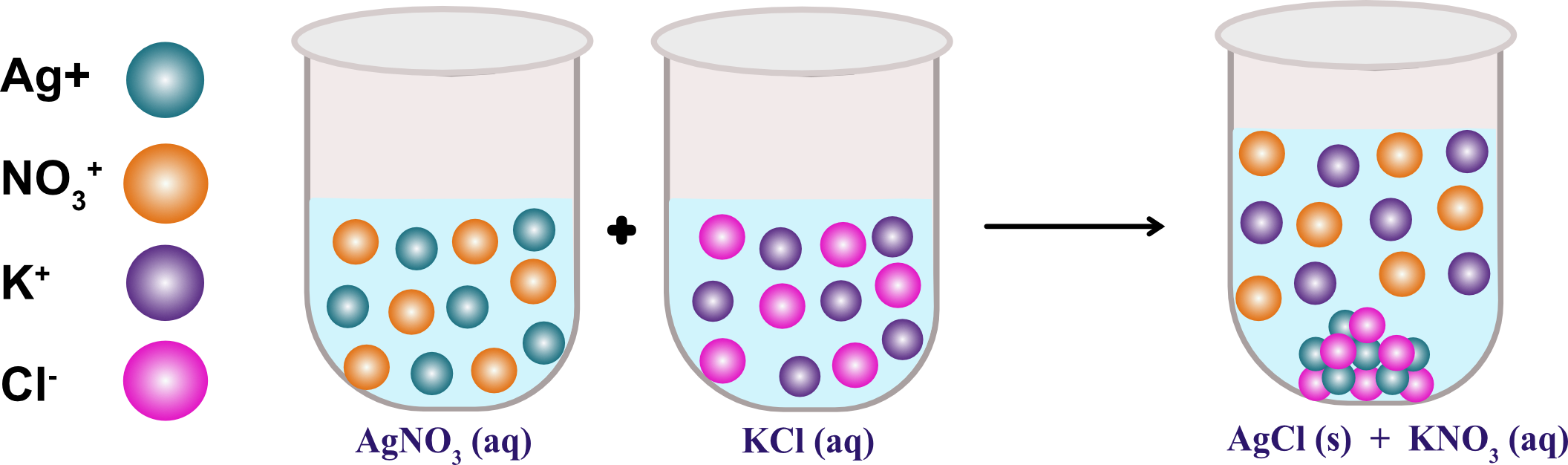
while this happens the rerrous ion loses one electron. from ferrous ion, the positive charge is increased by one unit. The conversion of ferrous sulphate to ferric sulphate is an oxidation reaction. The conversion or ferrous sulphate to ferric sulphate is ……. To prevent corrosion, a layer of zinc metal (galvanisation) is applied on iron sheets.ī. Due to corrosion of an iron a deposit of reddish substance (Fe 2O 3.H 2O) is formed on it. The rusting of iron is an oxidation process. To prevent rusting, a layer of zinc metal is applied on iron sheets. To prevent rusting, a laver of ……… metal is applied on iron sheets. (Oxidation, displacement, electrolysis, reduction, zinc, copper, double diplacement, decomposition)Ī. Maharashtra State Board Class 10 Science Solutions Part 1 Chapter 3 Chemical Reactions and EquationsĬhoose the correct option from the bracket and explain the statement giving reasons : Balbharti Maharashtra State Board Class 10 Science Solutions Part 1 Chapter 3 Chemical Reactions and Equations Notes, Textbook Exercise Important Questions and Answers. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
